David and I met after my long summer “vacation” in England. I guess I started work on TS06 yesterday Sept, 8th. Today, of course, we can expect semen to be all over the sidewalks of SF because Apple announce their iWatch today. How has this company managed to get such blatant world-wide free advertising?
Objective
Basically we are trying to make a smaller version of TS05b with the following improvements:
- Better ranging using the DW chip
- Better power utilization, maybe incorporating some on-board generation capabilities
- Smaller – with a view to a wrist worn device
There are a bunch of other little changes, but these are the fundamental ones.
Target Processor issues
So the first job was to figure out the plan of attack. The DW EVK uses an STM32F105 ARM chip, a Cortex-M3 core, as the processor. It has 256K flash, and 64K RAM and a maximum CPU speed of 72 MHz.
We are going to use a seriously more constrained processor – the Nordic nRF51822 BLE chip – because it has BLE. It is only a 32-bit ARM Cortex M0 processor with 256kB flash, and only 16kB RAM. The BLE stack consumes a lot of space and we have only about 180K left for our code. The CPU speed is 16 MHz. Let’s hope it can handle the interrupts.
So my major worry is that the DW stack soaks up a lot of RAM for buffers and so forth and we run out. So the first goal is to answer this question, and until we have an answer it makes developing anything more than minimal hardware a potential big waste of time.
Setting up Keil IDE – Day #2
I’ve spent a lot of time trying to set up Eclipse for the Nordic and it has been quite a struggle. I suceeded but I’m not feeling confident that it is stable – or that I could do it again, but maybe I’ll have to.
Unfortunately the propreitary system from Keil system costs $9K, but it looks like it has some decent libraries and a RTOS which could be useful. It would be good to evaluate the Keil system and decide if all those apparent advantages are real. Yet again David has worked some kind of magic that was way beyond my capabilities and located a system we can evaluate for an extended period. Our problem now is downloading it and trying it out. It consumes 5GB, takes 2 hours to download, and has totally maxed out the laptop I was going to use to evaluate it.
Installation
- Install mdk511a.exe When it finishes the Pack Installer will start up and you’ll notice that there are no Nordic devices. Quit the Pack Installer for now.
- Install Legacy support mdkcm510.exe
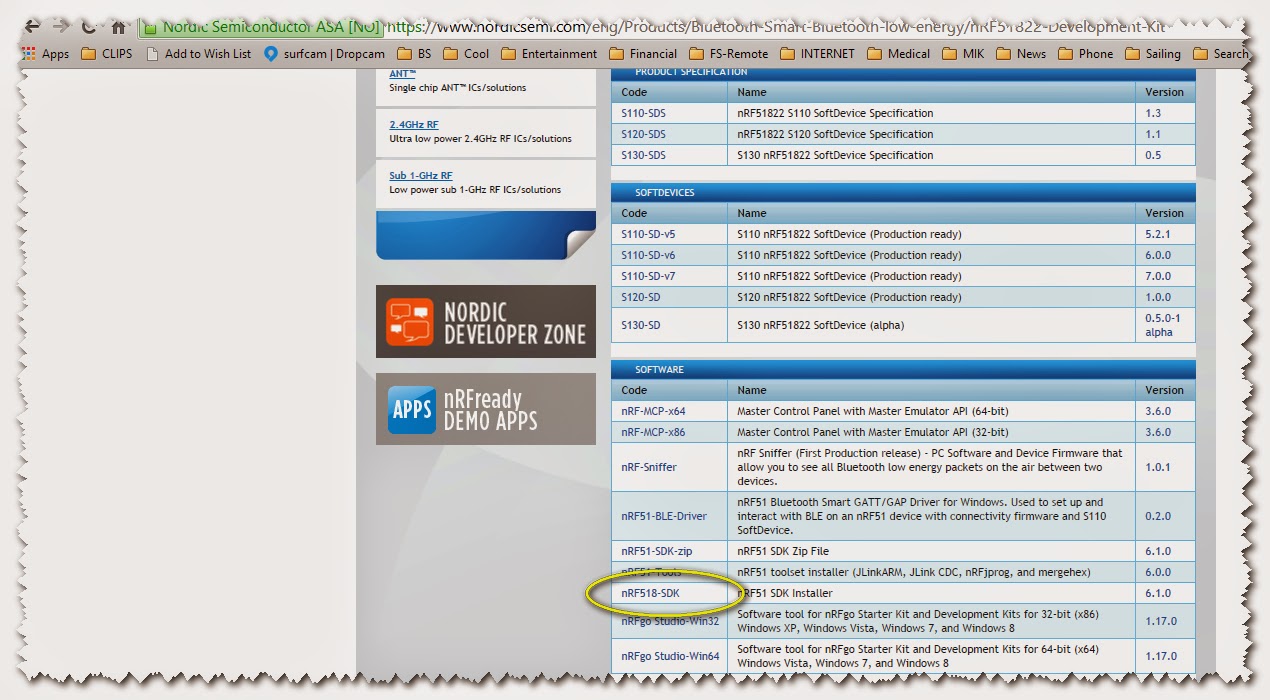
- Install nrf51_sdk_v6_1_0_b2ec2e6.msi from here.
making sure to include the nRF51822 examples,
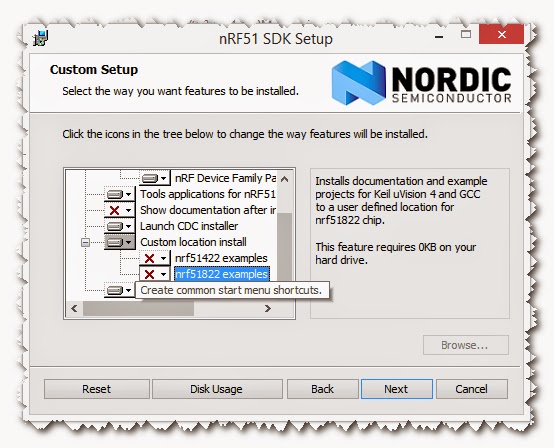
Make sure to include nRF51822 examples and make sure that the nRF Device Family Pack is going to be included
- and at the end accept the CDC OB CDC Driver package,
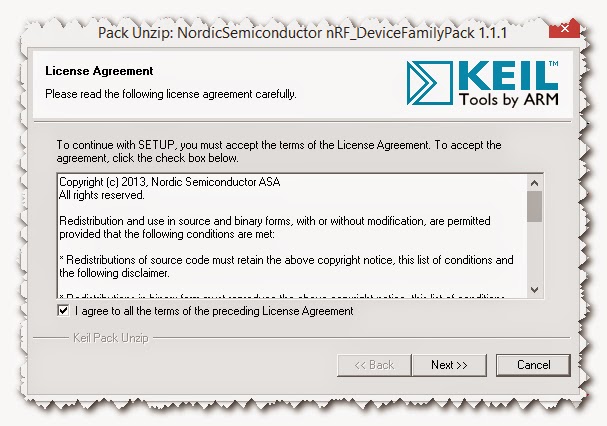
and accept the terms of the nRF_device Family Pack
- Start uVision and select Project/Manage/Pack Installer and make sure that the appropriate Nordic parts, and the STMF105 (DecaWave) are installed. If not install them.
- Now you have to make a valid license. To do that right-click run-as-administrator the new Keil iVision5 system you have just created.
-
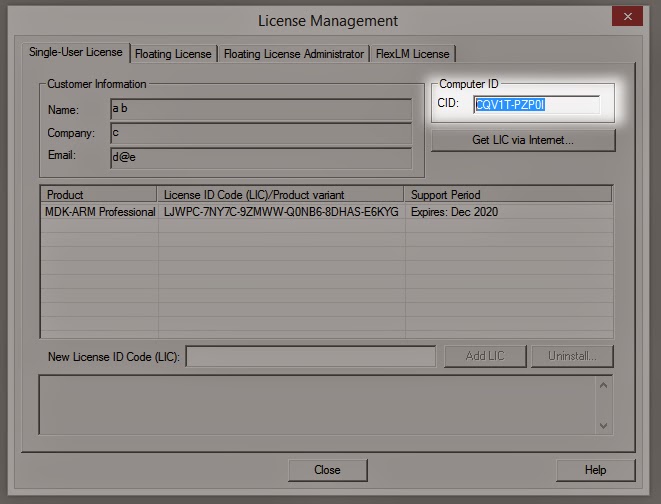
Click File/License Management
-
Copy the number out of the CID field
-
Disable Virus Checking, start the KeyGen, paste in the CID, and click generate.
- Copy the key that gets generated in the bottom field of the KeyGen into the New License ID Code field of the License Manager, and press Add Lic
- A license key will be generated and you can verify the expiry date.
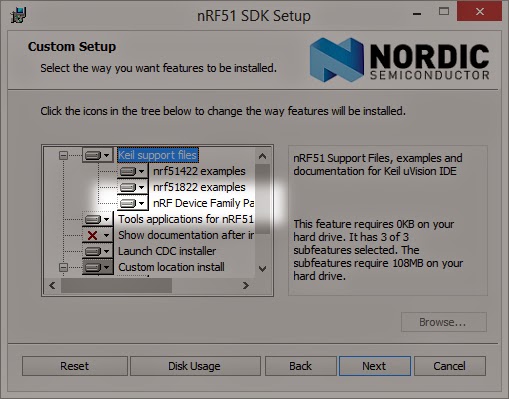
 |
| Not that there are no Nordic devices |





 When I was 10 years old, I had a crush on a girl named Mary. Mary knew my love was real—when I saw her, my mood ring would turn from a swirly green to a wavy, purplish-orange. As an adult, I now have more sophisticated tools—tools that measure information related to my health and fitness. I’ve traded in my mood ring for my Fitbit®, which I have on from the moment I leave the house to walk our dog, Tarot. Tarot has an activity monitor too, which tracks his location by GPS in case he runs off. And, at the end of the day, my wife compares my results to Tarot’s on her smartphone and decides who sleeps on the floor. Wearables have changed our lives.
When I was 10 years old, I had a crush on a girl named Mary. Mary knew my love was real—when I saw her, my mood ring would turn from a swirly green to a wavy, purplish-orange. As an adult, I now have more sophisticated tools—tools that measure information related to my health and fitness. I’ve traded in my mood ring for my Fitbit®, which I have on from the moment I leave the house to walk our dog, Tarot. Tarot has an activity monitor too, which tracks his location by GPS in case he runs off. And, at the end of the day, my wife compares my results to Tarot’s on her smartphone and decides who sleeps on the floor. Wearables have changed our lives.